 |
It is possible to extract the interatomic screening potential from experimental data [43], but this technique can just be applied to a limited number of atom types. More successfully is to perform a simplified quantum-mechanical calculation of the penetration of two atoms as suggested by Gombas [28]. Based on this method various interatomic potential have been calculated [88], [92] which mainly differ by the the atom models and the corresponding electron charge distributions of the atoms. Classical charge distributions such as those of Bohr [11], Thomas-Fermi [77], Lenz-Jensen [42], [49] and Moliére [58] have been used for the calculations as well as quantum-mechanical charge distributions derived by the Hartree-Fock approximation [23] for isolated atoms or by solid-state atomic models [47].
The interaction potential is calculated by assuming that the charge distributions of the atoms remain unchanged during the penetration of the electron spheres and that the charge distributions are spherically symmetric. The following contributions to the potential energy are considered.
Ziegler, Biersack and Littmark have performed potential calculations for a wide range of particle pairs (522 atoms pairs). They assumed solid state charge distributions for both particles, which were constructed by using a Hartree-Fock-Slater [23] method or in some cases by local density approximations for exchange and correlation effects as proposed in [59].
To be able to apply the solid state charge distribution to the semi quantum-mechanical model the real, anisotropic solid state charge distribution was transformed to a spherical symmetric distribution by averaging over all spatial directions.
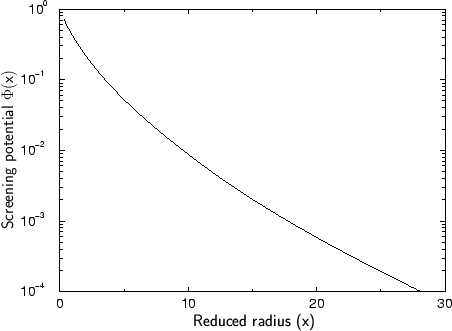
On the basis of their calculations they could extract an approximate analytical
expression for the dimensionless screening potential, the universal
screening potential (USP) ![]() plotted in Fig. 3.4
[92]. This potential should be suitable for arbitrary atom pairs.
plotted in Fig. 3.4
[92]. This potential should be suitable for arbitrary atom pairs.
Comparing the universal screening to the simulation results which were the base for the extraction of this analytical approximation function it can be said that the potential is approximately valid for almost all atom pairs. The deviation between the 522 calculated potentials and the fitted universal screening potential is approximately 18 % as long as the potential energy is above 2 eV. For lower potential energies the deviation is significantly larger which probably limits the use of the universal screening potential.
![]()
![]()
![]()
![]() Previous: 3.3.2 Nuclear Stopping Process
Up: 3.3.2 Nuclear Stopping Process
Next: 3.3.2.2 Average Nuclear Stopping
Previous: 3.3.2 Nuclear Stopping Process
Up: 3.3.2 Nuclear Stopping Process
Next: 3.3.2.2 Average Nuclear Stopping